
For Research Use Only. Not for human use.
Ultra Pure Peptides – Research Use Only – Raw Material Sourced from the USA
For Research Use Only. Not for human use.
Ultra Pure Peptides – Research Use Only – Raw Material Sourced from the USA
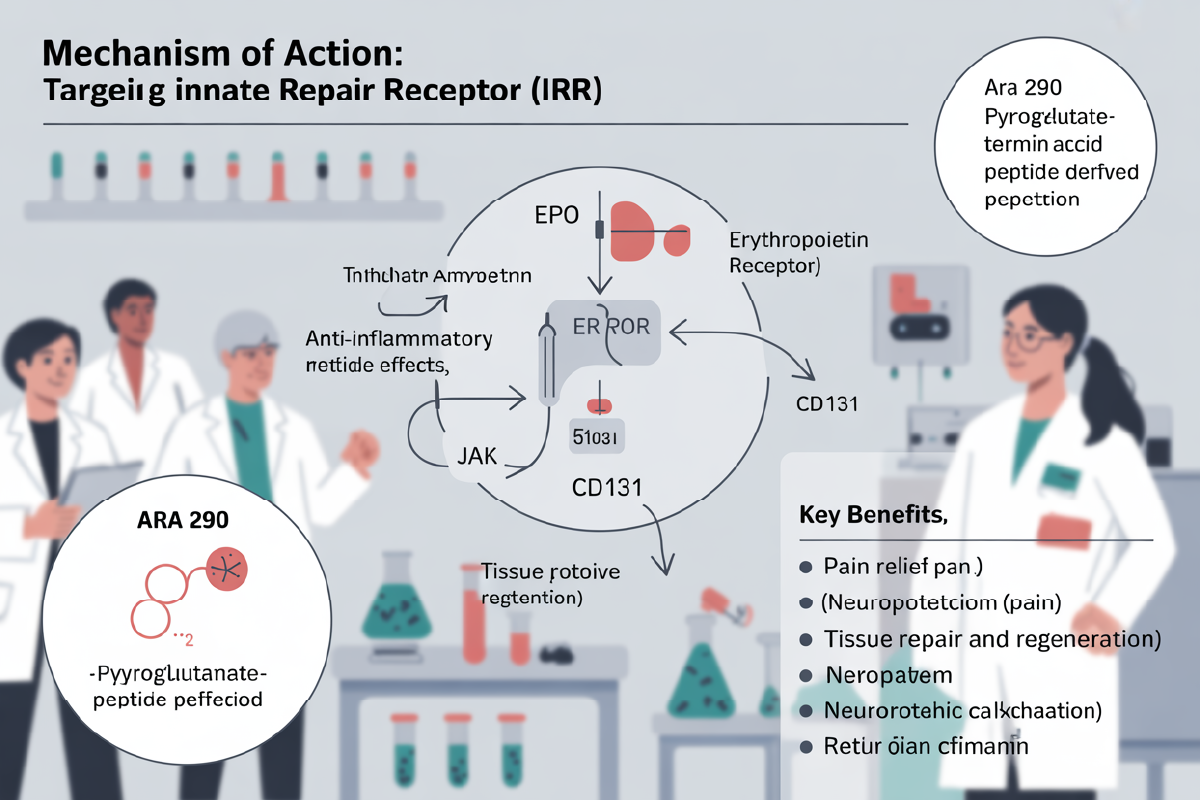
The ARA-290 is the engineered and non-erythropoietic peptide that can bind the innate repair receptor to reduce chronic inflammation and neuropathic pain. The main key benefits may include the small fiber neuropathy (like in sarcoidosis) for enhancing tissue repair, along with increasing insulin sensitivity. This can protect nerves without stimulating the red blood cell production.
The ARA 290 peptide can selectively activate the innate repair receptor, twhichis a complex of the EPO receptor and beta common receptor (CD131), as it bypasses the hematopoietic and the EPO receptor.
It can suppress the proinflammatory cytokines by modulating the macrophage activity and the microglial response as it reduces the inflammation in the tissue injury.
It promotes cell survival and repair by upregulating the anti-apoptotic pathways, as it supports angiogenesis. Also, it can be seen in wound healing models.
Neuropathic Pain Relief: It reduces the allodynia and hyperalgesiaby attenuating the spinal cord inflammation and possibly inhibiting the TRPV1, which channels the activity in the nociceptor, as it can be under investigation.
It improves the glucose metabolism, especially in the lipid profiles, through the indirect GH/IGF-1, as the modulation of the IRR can be mediated through tissue repair.
The clinical studies have shown that buy ARA-290 can reduce the pain symptoms, improve the nerve ffibreintegrity and enhance the functional measure in the affected patients.
In the rat models of the injury, the ARA, 290 can reduce the mechanical and the cold allodynia as it cansuppress the activation of the spinal microglia. It indicates the anti-inflammatoryeffects.
It’s a phase 2 study that shows how daily ARA-290 can improve the HbA1C and the lipid profiles in people with type 2 diabetes.
This trial was found in the neuropathic pain symptoms as it increases the corneal small nerve fibre. Suggesting it to be symptomatic and structural benefits.
The preclinical work suggests that ARA-290 can exert the protective effects in models of the inflammatory and ischemic injury that is also outside of the nervous system. It also includes the reduction of heart inflammation, protection in the injury models and suppression of theautoimmune nerve inflammation.
With some kind of exploratory research points to the improved microvascular integrity and tissue perfusion in the experimental models and small human studies (diabetic ocular condition with injury models).
Buy ARA-290 as it can have reduced inflammatory signalling that supports the structural preservation in the autoimmune encephalomyelitis model, as it suggests the potential neuroprotective applications.
The modulation of the innate immune cells and the prevention of Alzheimer’s type pathology is observed in the early experimental models, as it highlights the future research directions.
The ARA290 has not been widely approved for clinical use outside of research contexts; the most current human data can come from phase 2 or small pilot trials.
In various safety profiles, these settings may appear highly favourable. Asthere are no erythropoietic side effects along with tolerability that is compared to placebo for the long-term safety outside of the research indications.c
For most of the proposed applications (systemic inflammation and neurodegeneration), the evidence base can remain treclinical in the early stages.
The ARA-290 is a targeted tissue protective peptide that can be derived from the EPO as it activates the reparative signalling without stimulating the blood cell production. It can be supported by the research application in neuropathic pain and small fibre neuropathy. The broader experimental role in tissue protection and potential neuroprotective therapies.
https://www.mdpi.com/2077-0383/9/7/2225
https://www.ahajournals.org/doi/10.1161/res.135.suppl_1.Mo112
https://clinicaltrials.gov/study/NCT06626971
https://www.sciencedirect.com/science/article/abs/pii/S0014299925003644
Dr Parker is a molecular biologist; she specialises in therapeutic and regenerative medicine. With almost 10 years of experience in peptide research, Dr Parker has contributed to research on cellular health and muscle recovery.